Being trained in computational and organic chemistry, my research interest will, therefore, lie in the borderline between computational chemistry and organic chemistry. My future projects will aim at advancing knowledge on the structure, function and binding of molecules relevant to drug design. Synthesis of these molecules and fundamental aspects in this domain will be probed using computational chemistry techniques. Reaction mechanisms elucidation using Molecular dynamics simulations, Molecular Docking and Quantum mechanics methods will be performed to understand how different medically/chemically important receptors/enzymes function. Research in my group will lead to the discovery/design and synthesis of new bio-active molecules and/or catalyst.
Towards the synthesis of b-Carbolines and Spiro-oxindoles
Project Description:
we will make highly potent organic molecules which are key structural component in compounds with diverse bioactivity such as anticancer, antibiotic, immunosuppressant, anti-fungal, and phytotoxic property. These organic molecules can also be use as probes in chemical biology. The carbolines and indoles are derived from the amino acid tryptophan. The -Carboline and Spiro-oxindole moiety is a characteristic feature of several classes of naturally occurring alkaloids. Prominent amongst these are the Strynchnos, Curare, and Aspidosperma types. These alkaloids have been shown to possess diverse biological properties.
Goal
The goal of this work is to develop a new class of bio-active agents.
To reach this goal, we will address the following scientific questions: What are the specific chemical and physical parameters that dictate bioactivity of these organic molecules? How can these molecules be engineered to improve their bioactivity while minimizing toxicity? Computational work (in this group) will provide fundamental insights into interaction and binding of various selected organic molecules in the presence of appropriate receptors. The top performing molecular structures identified through computation will be synthesized (in this group) and their bioactivity – experimentally verified (via collaboration).
Design and Synthesis:
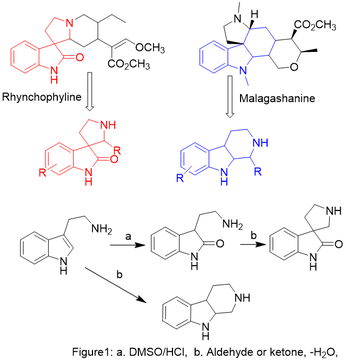
For the medicinal chemist, a biologically active natural product provides a useful template for designing novel molecules which can be used to probe the mechanism of the parent compound, or to develop drugs with improved pharmacological profiles. Inspiration for the design of this class of molecules came from an inspection of the structure of malagashanine, and rhynchophyline. As shown in figure 1, a tryptaminyl or tryptamine-like fragment can be found embedded in all 2 natural products. After a critical look, the Spiro-oxindole (in red) and -carboline (in blue) moieties emerged as the consensus structure for this drug discovery effort.
We will build -carboline and spiro-oxindole using a series of condensation reactions that involve intramolecular Mannich and Pictet-Spengler reaction. The precursors for these reactions are available from tryptamine, insatin or a tryptophan-derived oxindole and an aldehyde or ketone. All the reactions will be monitored using standard organic synthesis techniques such as thin layer chromatography (TLC) and/or high-performance liquid chromatography (HPLC). Proton and Carbon-13 NMR spectra will be recorded on the department’s NMR. Chemical shift will be reported relative to chloroform (CDCl3), methanol (d3-MeOH) and if necessary dimethyl sulfoxide (d6-DMSO). High-resolution mass spectra (HRMS) will be recorded at an appropriate center.
Academic Significance:
In this project, we will make use of simple condensation reactions to build bio-important molecules as shown in figure 1. The students will be able to make molecules with specific stereochemistry, use computational techniques to illustrate the reaction mechanism and rationalize the stereo-specificity. The student will also make use of 1D and 2D NMR techniques as a structural assignment tool. Therefore, students who have completed or are currently taking organic chemistry classes and those with ambitions of having a career in the pharmaceutical sector will find great interest in joining my group.
Project Description:
we will make highly potent organic molecules which are key structural component in compounds with diverse bioactivity such as anticancer, antibiotic, immunosuppressant, anti-fungal, and phytotoxic property. These organic molecules can also be use as probes in chemical biology. The carbolines and indoles are derived from the amino acid tryptophan. The -Carboline and Spiro-oxindole moiety is a characteristic feature of several classes of naturally occurring alkaloids. Prominent amongst these are the Strynchnos, Curare, and Aspidosperma types. These alkaloids have been shown to possess diverse biological properties.
Goal
The goal of this work is to develop a new class of bio-active agents.
To reach this goal, we will address the following scientific questions: What are the specific chemical and physical parameters that dictate bioactivity of these organic molecules? How can these molecules be engineered to improve their bioactivity while minimizing toxicity? Computational work (in this group) will provide fundamental insights into interaction and binding of various selected organic molecules in the presence of appropriate receptors. The top performing molecular structures identified through computation will be synthesized (in this group) and their bioactivity – experimentally verified (via collaboration).
Design and Synthesis:
For the medicinal chemist, a biologically active natural product provides a useful template for designing novel molecules which can be used to probe the mechanism of the parent compound, or to develop drugs with improved pharmacological profiles. Inspiration for the design of this class of molecules came from an inspection of the structure of malagashanine, and rhynchophyline. As shown in figure 1, a tryptaminyl or tryptamine-like fragment can be found embedded in all 2 natural products. After a critical look, the Spiro-oxindole (in red) and -carboline (in blue) moieties emerged as the consensus structure for this drug discovery effort.
We will build -carboline and spiro-oxindole using a series of condensation reactions that involve intramolecular Mannich and Pictet-Spengler reaction. The precursors for these reactions are available from tryptamine, insatin or a tryptophan-derived oxindole and an aldehyde or ketone. All the reactions will be monitored using standard organic synthesis techniques such as thin layer chromatography (TLC) and/or high-performance liquid chromatography (HPLC). Proton and Carbon-13 NMR spectra will be recorded on the department’s NMR. Chemical shift will be reported relative to chloroform (CDCl3), methanol (d3-MeOH) and if necessary dimethyl sulfoxide (d6-DMSO). High-resolution mass spectra (HRMS) will be recorded at an appropriate center.
Academic Significance:
In this project, we will make use of simple condensation reactions to build bio-important molecules as shown in figure 1. The students will be able to make molecules with specific stereochemistry, use computational techniques to illustrate the reaction mechanism and rationalize the stereo-specificity. The student will also make use of 1D and 2D NMR techniques as a structural assignment tool. Therefore, students who have completed or are currently taking organic chemistry classes and those with ambitions of having a career in the pharmaceutical sector will find great interest in joining my group.